
The number of protons influences the chemical behaviour of an element. For example, any atom that contains exactly 20 protons in its nucleus is an atom of calcium. 
The atomic number is equal to the number of protons in an atom's nucleus and determines which element an atom is. The number above the atomic symbol represents the atomic number. Some people attribute the first categorization to Alexandre-Emile Béguyer de Chancourtois.Ĭlose-up of how an element is presented in the periodic table (©2021 Let’s Talk Science). In Germany, Lothar Meyer also came up with an almost identical table later the same year. Dmitri Mendeleev, a Russian scientist, is usually credited with creating the first periodic table in 1869. The periodic table of the elements is a visual and logical way to organize all elements. This is because its natives gave silver gifts to the first European conquerors. Its name is from the latin name of silver, argentum. Other elements have been named for places, such as: ElementĪrgentina got its name from an element. Pierre and Marie Curie (research in radioactivity)ĭmitri Mendeleev (creator of the first periodic table) Greek word neos meaning ‘new’ or ‘different’Īs well for famous scientists such as: Element Greek word hydrargyrum meaning ‘silver water’ Many of the elements have their name or their symbol based in Latin or Greek. Some elements have even been discovered within your lifetime! Naming the Elements Scientists discovered many others during the 18th and 19th centuries. People have known about some elements such as gold, silver, iron, mercury and tin for hundreds of years. Matter that is composed of only one type of atom is called an element. Open Professional Learning × Close Professional Learning 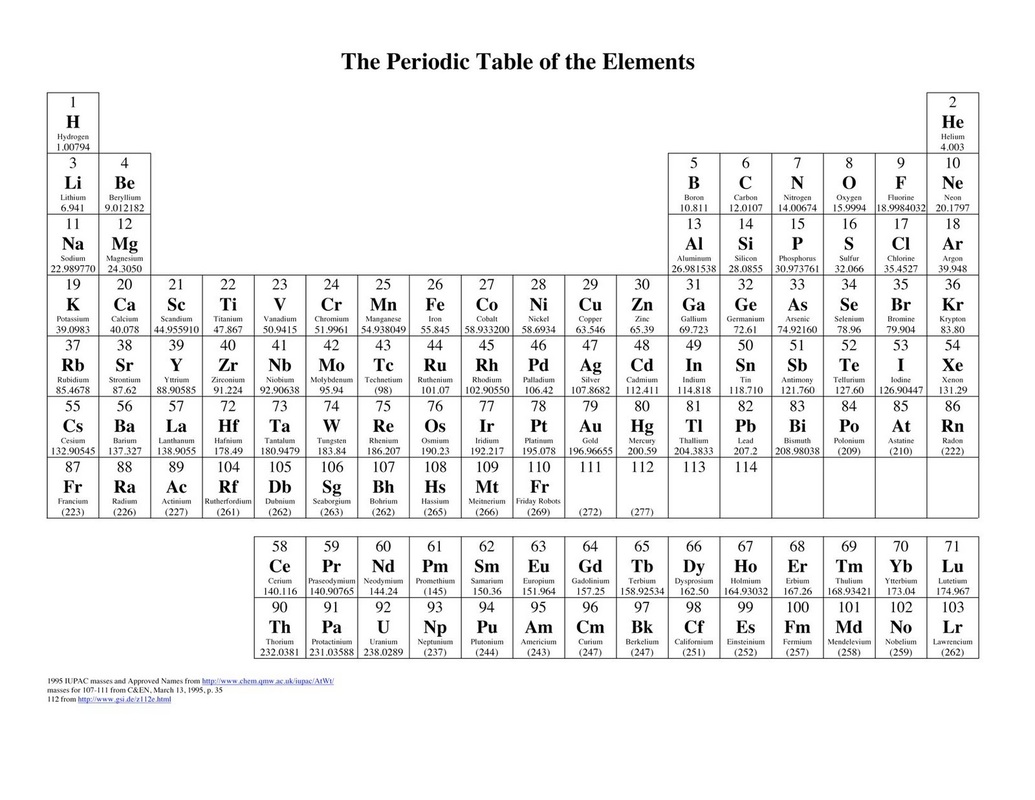
Elements can be categorized into s-block, p-block, and d-block elements based on their highest-energy orbital.Open Educational Resources × Close Educational Resources While there are many orbital shapes that are possible, the MCAT will focus on three particular orbitals denoted as s, p, and d. Some elements, like noble gases, already have a full shell.Įach shell is composed of orbitals: regions of certain shapes in which electrons are likely to be found. Each atom is at its most energetically stable when its valence shell is full of electrons. These electrons are responsible for the reactivity and individual properties of atoms. Valence electrons are electrons on the outermost shell of an atom. This outermost shell is also called the valence shell. The outermost electron shell, or highest-numbered electron shell, is located the furthest. As a general rule, electron shells of a higher number are located further from the nucleus. Thus, elements in the first row of the periodic table have one electron shell, elements in the second row have two electron shells, and so forth. The number of concentric shells is determined by the group number of an atom. While these shells are not physical structures, they are useful in visualizing the spaces that electrons may occupy. When “filling” in an atom, electrons are placed in concentric shells surrounding an atom. For ions, or particles with a net positive or negative charge, the number of electrons may not be equal to the atomic number of the element. Thus, if a particle has zero charge, the number of electrons and number of protons must be equal). (Why is this? Recall that the charge of one electron is equal to the charge of one proton. For most atoms, or neutrally charged particles, this number of electrons is equal to the atomic number of the element. Valence electrons play an important role in chemical reactions and bonding.Īn electron configuration is written by determining the number of electrons that are present in the species at hand. The electrons in the outer shell of an atom are called valence electrons. Electrons can be arranged in shells around an atom. In other words, when comparing the mass of a proton or neutron to an electron, the mass of an electron is negligible. Groups of protons and neutrons-collectively referred to as nucleons-form the nucleus of an atom.Īn electron is a subatomic particle that contains a negative charge (-1 e) and has a mass that is 1823x smaller than that of a proton, or 1/1823 atomic mass units. A neutron is a subatomic particle that contains no charge and has a mass of one AMU. “e” refers to the fundamental unit of charge and one atomic mass unit (AMU). 
A proton is a subatomic particle that contains a positive charge (+1 e). Protons, neutrons, and electrons are the subatomic particles that make up atoms. This is the result of the structure of individual atoms, which are composed of varying subatomic particles. Atoms are the smallest unit of matter that can retain a unique identity.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
